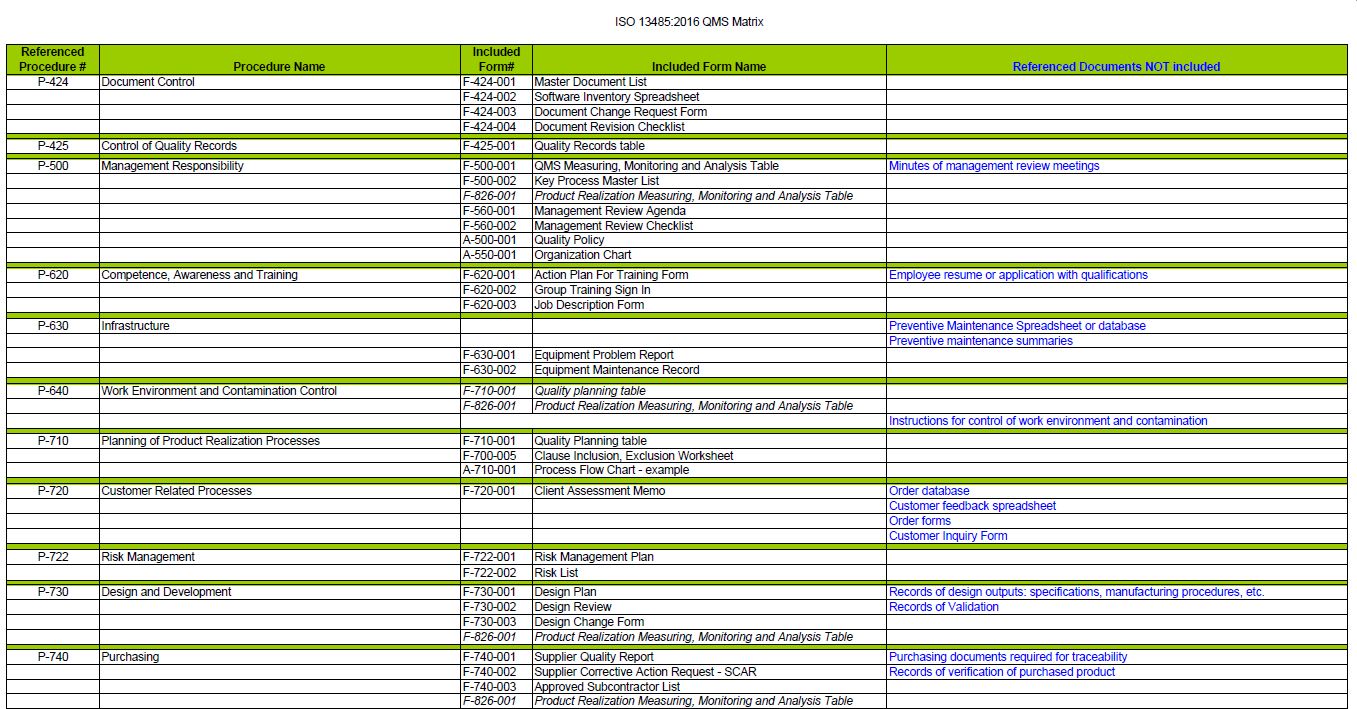
Evidence for visible top management leadership, commitment and quality management action must be looked for. The auditors must use a great deal of discretion and therefore must be careful and thoughtful prior to establishing a deficiency against a requirement. For other clauses of the standard, the phrase such as ‘documented procedures’ is used to specify that a process, a method, a system, a work instruction, or an arrangement be documented. The auditors are expected to keep in mind that the standard requires six (6) mandatory procedures, such as with clauses 4.2.4, 4.2.5, 8.2.4, 8.3, 8.5.2, and 8.5.3. You will see questions on the checklist that refer to the standard, and for each clause, provisions are made for additional questions. The checklist is best used by trained and practicing auditors to evaluate or assess Quality Management Systems requirements based on the standard. The ISO 13485:2016 Internal Audit Checklist This checklist is based on the information provided in the release of the ISO 13485:2016 international standard. 
During assessment of each requirement, auditors record the status of the evaluation by indicating in the right hand column a Yes - for Acceptable Condition or No - for Deficient Condition The bold numbers and tittles used in the first two columns of the checklist indicate the “Requirements” and may be referred to on nonconformity reports prepared by the auditor. 
©2016 1 The ISO 13485:2016 Internal Audit Checklist This checklist is based on the information provided in the release of the ISO 13485:2016 international standard.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
